 H2O (g)
H2O (g)
Heat of formation is defined as the enthalpy change when one mole of a compound is formed from the elements in their stable states. For example, the heat of formation of water vapor is defined by the reaction:
H2 (g) + 1/2 O2 (g)  H2O (g)
H2O (g)
Even though we generally want the coefficients of balanced reactions to be whole numbers, the definition of heat of formation specifies one mole of the product, so we leave the 1/2.
This reaction releases 241.8 kJ of heat when one mole of water vapor is produced. The heat of formation of water vapor is -241.8 kJ. (Why is this negative? - heat is released!)
The enthalpy change of a reaction (heat of reaction) can be calculated from heat of formation data. The heat of reaction (which for a combustion reaction is the heat of combustion) is calculated as:
Heat of reaction = [the sum of all heats of formation of all products] - [the sum of all heats of formation of all reactants]
Let's do an example.
The combustion of methane gas is represented by the reaction:
CH4 (g) + 2 O2 (g)  CO2 (g) + 2 H2O (g)
CO2 (g) + 2 H2O (g)
The calculation for the heat of combustion for methane is then:
heat of combustion =
= [ -393.5 kJ + 2x(-241.8 kJ)] - [-74.8 kJ + 2x(0 kJ)]
= - 802.3 kJ
So, the heat of combustion, that is, the heat of reaction, is - 802.3 kJ per mole of methane.
Notice that the value is negative, so the reaction gives off heat as expected for a combustion reaction.
Now you're ready to do the same for cellulose and it's derivatives.
Since cellulose is a large molecule (it's a polymer), we will do an approximate calculation
using a small molecule to represent the "monomer" of cellulose. For cellulose we will choose glucose
(can you explain why?). Glucose (and the other "monomers" that you'll need later) are given below.
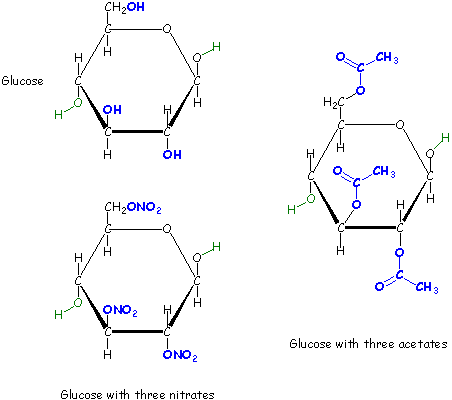
To do the calculation you first need to balance the reaction. Since glucose (and cellulose) contain only carbon, hydrogen and oxygen, we will assume that the products are only carbon dioxide gas and water vapor.
Have you finished balancing the reaction? OK.
Now that you have a balanced reaction, you can use the heats of formation to calculate the heat of combustion for cellulose.
The data needed to do the calculations can be found here.
When you're done you can continue on and check your answer for glucose.
No Peeking! Wait until you're done with the calculation.

|

|

|