|
|
There was a time long past when the only rubber we had was natural rubber latex, polyisoprene. Straight out of the tree, natural rubber latex isn't good for much. It gets runny and sticky when it's warm, and it gets hard and brittle when it's cold. Tires made out of it wouldn't be much good unless one lived in some happy land where the temperature was seventy degrees all year 'round.
A long time ago - in 1839 to be exact - one person trying to make rubber more useful was Charles Goodyear, a tinkerer and inventor, and not a very successful one at that point. While goofing around in his kitchen with a piece of fabric coated with a mixture of rubber, sulfur, and a little white lead, he accidentally laid it on a hot stove top. It began sizzling like a mass of really smelly bacon or (strangely enough) burning rubber. Wouldn't you know, when he took a look at this mass of rubber, he found it wouldn't melt and get sticky when it was heated, nor would it get brittle when he left it outside overnight in the cold Massachusetts winter. He called his new rubber vulcanized rubber.
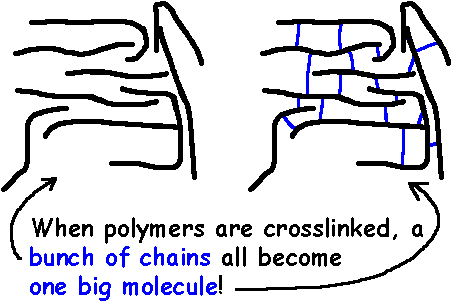
What had happened here? What did the sulfur do to the rubber? What it did was it formed bridges that tied all the polymer chains in the rubber together. These are called crosslinks. You can see this in the picture below.

Bridges made by short chains of sulfur atoms tie one chain of polyisoprene to another, until all the chains are joined into one giant supermolecule. An object made of a crosslinked rubber is in fact one single molecule. A molecule big enough to pick up in your hand!
Wow!
These crosslinks tie all the polymer molecules together. Because they're tied together, when the rubber gets hot, they can't flow past each other, nor around each other. This is why it doesn't melt. Also, because all the polymer molecules are tied together, they aren't easily broken apart from each other. This is why Charles Goodyear's vulcanized rubber doesn't get brittle when it gets cold.
Take a look at the bigger picture. The drawing on the right shows the difference between a lot of single uncrosslinked polymer chains, and a crosslinked network.
But rubber isn't the only thing that can be crosslinked. Plastics are also made stronger by crosslinking. Crosslinked polymers are usually molded and shaped before they are crosslinked. Once crosslinking has taken place, usually at high temperature, the object can no longer be shaped.
Because heat usually causes the crosslinking which makes the shape permanent, we call these materials thermosets. They are different from thermoplastics, which aren't crosslinked and can be reshaped once molded. The first thermoset was again polyisoprene. The more sulfur crosslinks you put into the polyisoprene, the stiffer it gets. Lightly crosslinked, it's a flexible rubber. Heavily crosslinked, it's a hard thermoset.
Crosslinking makes both elastomers and plastics stronger, but there's a problem. Because crosslinked materials don't melt, it's very hard to recycle them. One answer to this problem is to create crosslinks that can be reversed, or undone, believe it or not. One family of materials using reversible crosslinks is thermoplastic elastomers

|
Return to Level Three |

|
Return to Main Page |