

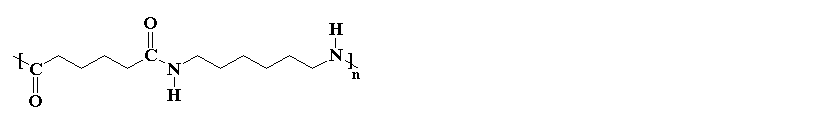
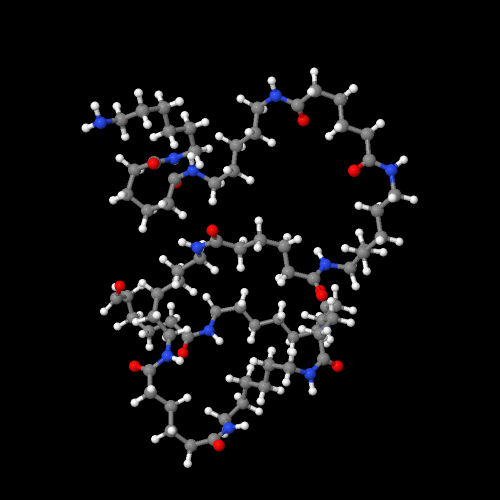
The 3D model is an image of the pdb model you can view by clicking here or you can just click on the image itself. Either way, be sure to close the new window that opens up with the 3D model in it when you are ready to come back here.
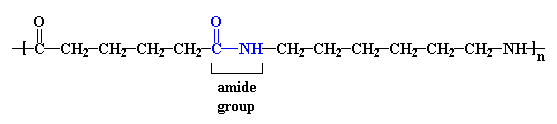
Nylons are also called polyamides, because of the characteristic
amide groups in the backbone chain. Proteins,
such as the silk nylon was made to replace, are also polyamides. These amide groups
are very polar, and can hydrogen bond with each other. Because
of this, and because the nylon backbone is so regular and symmetrical,
nylons
are often crystalline, and make very good fibers.
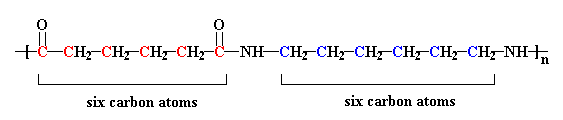
The nylon in the pictures on this page is called nylon 6,6, because each repeat unit of the polymer chain has two stretches of carbon atoms, each being six carbon atoms long. Other nylons can have different numbers of carbon atoms in these stretches.
Nylons can be made from diacid chlorides and diamines. Nylon 6,6 is made from the monomers adipoyl chloride and hexamethylene diamine.
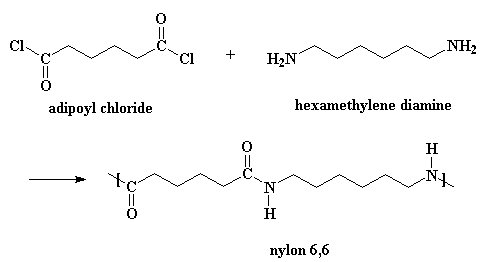
This is one way of making nylon 6,6 in the laboratory. But in a nylon plant, it's usually made by reacting adipic acid with hexamethylene diamine:
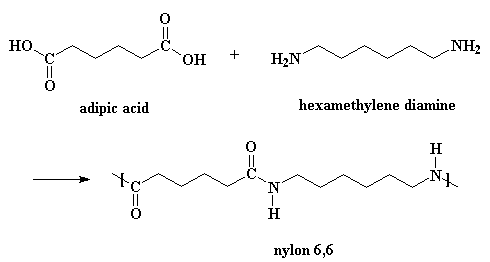
The model on the left is an image of the adipic acid pdb model you can view by clicking here or you can just click on the image itself. The second image is of the diamine monomer. Either way, be sure to close the new windows that open up with the 3D model in it when you are ready to come back here.


If you want to know how this condensation polymerization works, click here.
Another kind of nylon is nylon 6. It's a lot like nylon 6,6 except that it only has one kind of carbon chain, which is six atoms long.
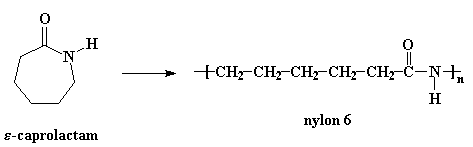
The image below is of the caprolactam pdb model you can view by clicking here or on the image.

A family of very different nylons with much better properties have their own page at aramids.
| Other polymers used as plastics include: | Other polymers used as fibers include: |
| Polypropylene | Polypropylene |
| Polyesters | Polyesters |
| Polystyrene | Polyethylene |
| Polycarbonate | Kevlar and Nomex |
| PVC | Polyacrylonitrile |
| Polyethylene | Cellulose |
| Poly(methyl methacrylate) | Polyurethanes |

|
Return to Level Two Directory |

|
Return to Macrogalleria Directory |
