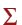 NiMi, where Ni is the number of molecules of weight Mi and the symbol
NiMi, where Ni is the number of molecules of weight Mi and the symbol 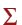 (it's one of those cool symbols that mathematicians use to impress people) tells you to sum up all the products of the individual NiMi.
(it's one of those cool symbols that mathematicians use to impress people) tells you to sum up all the products of the individual NiMi. Now that you have a general idea of what molecular weight means for a polymer, you can calculate the number average and weight average molecular weights for a small sample. What is the information that you have available? Usually you will have information about the molecular weight distribution obtained from Size Exclusion Chromatography. The data will look something like this:
| Number of Molecules | Mass of each Molecule |
| 1 | 800,000 |
| 3 | 750,000 |
| 5 | 700,000 |
| 8 | 650,000 |
| 10 | 600,000 |
| 13 | 550,000 |
| 20 | 500,000 |
| 13 | 450,000 |
| 10 | 400,000 |
| 8 | 350,000 |
| 5 | 300,000 |
| 3 | 250,000 |
| 1 | 200,000 |
Remember that this is a very small sample used to illustrate the concept. A real sample may have billions and billions of molecules. (Thanks Carl Sagan!!)
The number average molecular weight is the total weight of the sample divided by the number of molecules in the sample. What's the total weight of the sample? First find the total weight of each type (or weight) of polymer. This means that you multiply the weight of the molecule by the number of molecules of that weight. For example, there are 13 molecules with a weight of 550,000 so the total weight of molecules with a weight of 550,000 is 13 x 550,00 or 7,150,000. (This seems like a really big number, but remember size DOES matter with polymers: these are really big molecules.) If you do this for all the types (or weights) of molecules and add the weights together you have the total weight of the sample. In mathematical form, the total mass is 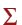 NiMi, where Ni is the number of molecules of weight Mi and the symbol
NiMi, where Ni is the number of molecules of weight Mi and the symbol 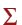 (it's one of those cool symbols that mathematicians use to impress people) tells you to sum up all the products of the individual NiMi.
(it's one of those cool symbols that mathematicians use to impress people) tells you to sum up all the products of the individual NiMi.
The data will look like this:
| Number of Molecules, Ni | Mass of Each Molecule, Mi | Total Mass of Each Type of Molecule, NiMi |
|---|---|---|
| 1 | 800,000 | 800,000 |
| 3 | 750,000 | 2,250,000 |
| 5 | 700,000 | 3,500,000 |
| 8 | 650,000 | 5,200,000 |
| 10 | 600,000 | 6,000,000 |
| 13 | 550,000 | 7,150,000 |
| 20 | 500,000 | 10,000,000 |
| 13 | 450,000 | 5,850,000 |
| 10 | 400,000 | 4,000,000 |
| 8 | 350,000 | 2,800,000 |
| 5 | 300,000 | 1,500,000 |
| 3 | 250,000 | 750,000 |
| 1 | 200,000 | 200,000 |
Total Mass = 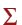 NiMi = 50,000,000 NiMi = 50,000,000 |
||
To get the number average molecular weight you divide the total weight of the sample by the total number of the molecules. How do you get the total number of molecules? Try 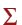 Ni (oh no! - you're starting to think like a mathematician!). In this case
Ni (oh no! - you're starting to think like a mathematician!). In this case 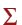 Ni = 100. The number average molecular weight for this sample is then
Ni = 100. The number average molecular weight for this sample is then 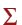 NiMi/
NiMi/ 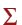 Ni = 50,000,000/100 = 500,000. Again, it's a really big number because polymers are really big molecules!
Ni = 50,000,000/100 = 500,000. Again, it's a really big number because polymers are really big molecules!
The calculation of the weight average molecular weight requires that you know the Weight Fraction, Wi, of each type of molecule (or the fraction of the total weight represented by each type of molecule). You don't know the weight fractions from the original data, but you can calculate them. So, what is the weight fraction? Like any fraction, it's the amount of the thing of interest divided by the total amount. For weight fraction of one type of molecule, it is the weight of that type of molecule divided by the total weight of the sample. Or, expressed in mathematical form, it is NiMi/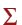 NiMi. Notice that you have already calculated both NiMi and
NiMi. Notice that you have already calculated both NiMi and 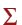 NiMi when you calculated the number average molecular weight, so you just need to do the division. The weight average molecular is then calculated by taking the sum
NiMi when you calculated the number average molecular weight, so you just need to do the division. The weight average molecular is then calculated by taking the sum 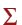 WiMi. When you add this information to the data table, it looks like this:
WiMi. When you add this information to the data table, it looks like this:
| Number of Molecules | Mass of Each Molecule | Total Mass of Each Type of Molecule |
Weight Fraction Type of Molecule |
|
|---|---|---|---|---|
| (Ni) | (Mi) | (NiMi) | (NiMi/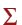 NiMi) NiMi) |
(WiMi) |
| 1 | 800,000 | 800,000 | 0.016 | 12,800 |
| 3 | 750,000 | 2,250,000 | 0.045 | 33,750 |
| 5 | 700,000 | 3,500,000 | 0.070 | 49,000 |
| 8 | 650,000 | 5,200,000 | 0.104 | 67,600 |
| 10 | 600,000 | 6,000,000 | 0.120 | 72,000 |
| 13 | 550,000 | 7,150,000 | 0.143 | 78,650 |
| 20 | 500,000 | 10,000,000 | 0.200 | 100,000 |
| 13 | 450,000 | 5,850,000 | 0.117 | 52,650 |
| 10 | 400,000 | 4,000,000 | 0.080 | 32,000 |
| 8 | 350,000 | 2,800,000 | 0.056 | 19,600 |
| 5 | 300,000 | 1,500,000 | 0.030 | 9,000 |
| 3 | 250,000 | 750,000 | 0.015 | 3,750 |
| 1 | 200,000 | 200,000 | 0.004 | 800 |
Weight Average Molecular Weight = 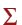 WiMi = 531,600 WiMi = 531,600 |
||||
Notice that the number average and the weight average molecular are not the same. The distribution of molecular weights in a polymer sample is often described by the ratio of the weight average molecular weight to the number average molecular weight. In this case the ratio is 531,600/500,000 = 1.063. This ratio is the Polydispersity Index (or PDI).

|
Return to the Molecular Weight Page |

|
Return to Macrogalleria Directory |