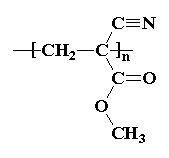
The model on the right above is an image of the pdb model
you can view by clicking here or you can just click on the image itself.
Either way, be sure to close the new window that opens up
with the 3D model in it when you are ready to come back here.
I once thought incorrectly that polycyanoacrylates were the only synthetic polymers I know of that play a significant role in great works of literature. In Kurt Vonnegut's novel Breakfast of Champions, our hero Kilgore Trout, the world's worst sci-fi writer, inadvertently gets his feet coated in a thick layer of a polycyanoacrylate, and he has to go to the emergency room in order to get the stuff chiseled off.
What Kilgore Trout ran up against was one of the more useful properties of polycyanoacrylates. Namely, they're great adhesives, so good in fact that they're used as superglues. And as poor Kilgore found out, they're very effective at bonding skin. Just ask anyone who has every stuck his of her fingers together with superglue and this will be verified.
You may be asking why these polycyanoacrylates make such great adhesives. Part of it is that they're really fast drying. Want to know how this works? Well, okay, I'll tell you. You see, the tube of wonderglue you buy in the store isn't a polycyanoacrylate at all. It's a tube full of a cyanoacrylate monomer, like this methyl cyanoacrylate:
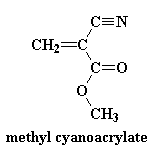
The model on the right above is an image of the pdb model
you can view by clicking here or you can just click on the image itself.
Either way, be sure to close the new window that opens up
with the 3D model in it when you are ready to come back here.
When you squirt this monomer onto whatever it is you want to glue, it polymerizes by anionic vinyl polymerization. Water from the air or trace amounts of moisture on the surface of that which you're gluing acts as the initiator.
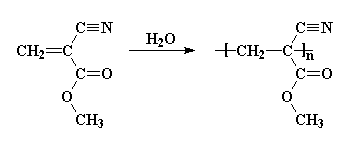
This polymerization takes place within seconds to give you the polycyanocrylate, poly(methyl cyanoacrylate) in the example in the picture. That's the same polymer in the pictures at the top of this page, but other alkyl cyanoacrylates can be used, too, like butyl cyanoacrylate and octyl cyanoacrylate.
Polycyanoacrylates have another useful property. They're non-toxic. Let's think about this: They bond skin, plus they're non-toxic. What could you do with something like that? How about using it instead of needle and thread to close up wounds? Some doctors are also trying to use polycyanoacrylates as glues to repair eyeball parts, like corneas and retinas. In addition, some people are testing films of polycyanoacrylates for use as synthetic skin to use in skin grafts for treating severe burns.
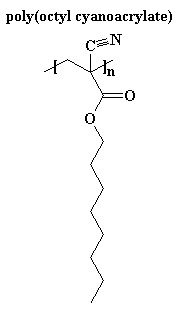
Usually for medical uses we use cyanoacrylates with longer alkyl ester groups than you find in super glues. A good example is poly(octyl cyanoacrylate), shown on the right. This is because polycyanoacrylates with short alkyl groups, like methyl groups, can irritate tissues. But the long chain polymers don't have this problem.
One last thing...remember when I said that I had thought incorrectly that polycyanoacrylates were the only polymers to play a big part in a literary work? I was incorrect because it turns out that in Gravity's Rainbow by Thomas Pynchon, a polymer which is not given a proper chemical name but sounds an awful lot like a polyimide of some sort plays a big part in the action of the novel.

|
Return to Level Two Directory |

|
Return to Macrogalleria Directory |
