 |
Keywords:
|
There are three sections to the metathesis page, so we're going to give you this handy index to help you navigate:
- What Is Olefin Metathesis?
- Acyclic Diene Metathesis Polymerization
- Ring Opening Metathesis Polymerization
What Is Olefin Metathesis?
In other pages on our site we've talked an awful lot about making polymers from monomers with carbon-carbon double bonds. Remember all our talk of vinyl polymers? Those polymerizations involve the double bond forming two new single bonds; the double bond disappears during the reaction.
But vinyl polymers don't often have double bonds in the polymer backbones (unless one monomer is butadiene, maybe). Now we're going to talk about taking monomers with double bonds, and using them to make polymers with double bonds in the backbone chain, without loss of the double bonds. We call these polymers polyalkenamers. One way to make polyalkenamers to use a nifty reaction called "olefin metathesis."
Olefin is an old fashioned word, kind of like betwixt or forsooth. An olefin is the same thing as an alkene; that is, a molecule with a carbon-carbon double bond. Olefin metathesis is (of course) a reaction involving olefins. Two olefins, to be exact. It seems counter-intuitive but here's what they do: the double bond carbons change partners, to form two new olefins, just like in the little picture here:
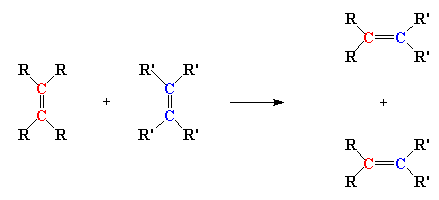
Neato, huh? But it isn't really obvious that this reaction can be used to make polymers. In fact, this reaction was discovered in the 1920s, and it wasn't until about fifty years later that scientists figured out a way to make polymers out of it. And it wasn't until around twenty years even later still that anyone made anything useful with the reaction. So turning this reaction into a polymerization took some clever scheming on the part of those polymer chemists. But succeed they did! And in fact, they came up with two ways of using olefin metathesis to make polymers.
TWO GREAT POLYMERIZATIONS,
ONE LOW PRICE!
So let's take a look at each of them, why don't we? That way we can understand how each one is different, how each one works overall, and how each can be used to make entirely different families of polymers, all with double bonds in their backbones.
Metathesis Polymerization #1:
Acyclic Diene Metathesis Polymerization
The two different polymerizations
are called ring opening metathesis polymerization, or ROMP, and acyclic diene metathesis
polymerization, or ADMET. We'll talk about ADMET first because it's simpler,
even though ROMP has been more thoroughly researched. In ADMET we start with
an acyclic diene (surprise, surprise) such as 1,5-hexadiene, and end up
with a polymer with double bonds in the backbone chain at regular repeat sequences.
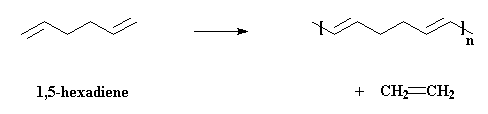
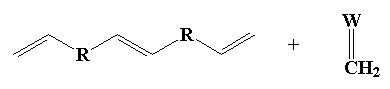
There's also the by-product, ethylene gas. Loss of ethylene helps drive the reaction to high molecular weight polymer (think: thermodynamics). How does metathesis make this possible? I'll show you how, using the example of two molecules of 1,5-hexadiene reacting together:
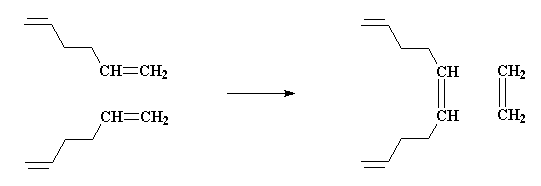
Ok, let's start with the first step, although it's actually the second one. The first involves formation of the metal complex that actually does the initiation step. Anyway, click below to see the metal carbene (carbon double bond to the metal catalyst) approach and bond to the terminal alkene (vinyl).
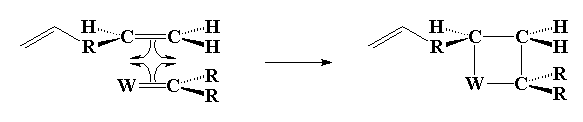
Click here or on the image above to see a movie of the first step.
The second step involves transfer of carbon groups:
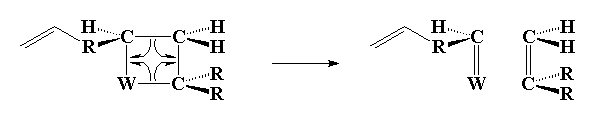
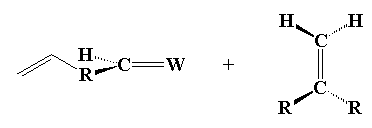
Click here or on the image above to see a movie of the second step.
And the third step with new complex formation:

Click here or on the image above to see a movie of the third step.
And the fourth and last steps with carbon group exchange completed:
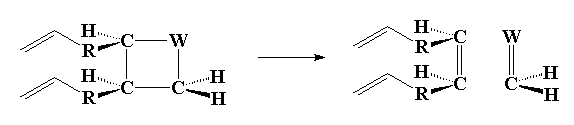
Click here or on the image above to see a movie of the last step.
Click here to see a movie of all steps of this reaction together.
And of course the double bonds at the ends of the new molecule can have a little metathesis magic done on them as well, and in this way the polymer grows. The cis- isomer of the product formed is shown above, but usually both cis- and trans- isomers can form, and there's usually room for both in the polymer that we end up with. That's part of what makes this process so magical. The polymer formed has both a unique structure and unique properties. Maybe you can find a new use for such novel materials?
Metathesis Polymerization #2:
Ring Opening Metathesis Polymerization
We can get polymers from cyclic olefins, too. Look at the picture below, and you see that a cyclic olefin, cyclopentene in this case, is used to make a polymer which doesn't have cyclic structures in its backbone.
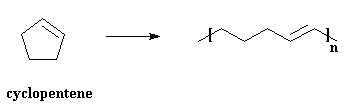
This is why we call it "ring-opening metathesis polymerization". Makes sense. ROMP is, believe it or not, used to make some useful products. A nifty little molecule, norbornene, is polymerized by ROMP to get polynorbornene. It's a kind of rubber used to make auto parts, the little body-mount parts that keep things from vibrating and such.
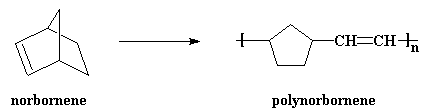
We can also make a polymer from a similar molecule using ROMP. That molecule is endo-dicyclopentadiene. When we polymerize it we get a polymer with a cyclic olefin in a pendant group. This polymer is polydicyclopentadiene, of course.

This pendant olefin can be reacted to crosslink the polymer, probably by traditional vinyl polymerization of some sort. The polymer is a hard plastic that is used to make satellite dish antennae, body parts for snowmobiles, and other things that are generally used outside. You don't want to use this stuff inside, because there's always a little bit of endo-dicyclopentadiene that hasn't reacted left trapped in the polymer. This is one malodorous monomer, and it doesn't take much to stink up a room.
Want to see all these nifty ROMP monomers and polymers in 3-D?
Click here!
How this happens takes some explaining. It's kind of complicated. It starts off with an initiation step.
Ok, kids what do we need for an initiation step? Maybe, an initiator? The initiator in this case is usually a compound with a carbon atom double bonded to a metal atom, like tungsten or molybdenum. The catalysts often look like this, but we're going to simplify the drawing.
So the initiator reacts with a molecule of cyclopentene in a metathesis reaction like we've been seeing, to get a molecule with a carbon-carbon double bond at one end, and a carbon-metal double bond at the other.

That carbon-metal double bond can react with another molecule of cyclopentene in the exact same way:

And in this way the polymer grows, until we get this:
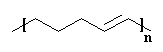

|
Return to Level Four Directory |

|
Return to Macrogalleria Directory |